SEPTEMBER 5, 2016: NOX66 TECHNOLOGY EXPLAINED by Dr Graham Kelly
Here is an obvious question:
Idronoxil was tested in over 400 cancer patients over a 10-year period. In the end, it didn’t work in enough patients to warrant the investment to continue to bring it to market. So why do we think we can make it work now?
Short answer:
The way the drug was used previously led to it facing hurdles that were not fully understood at the time. In essence, idronoxil belongs to a class of chemicals that the body subjects to a process of detoxification involving 2 steps. The first step inactivates the drug; the second step reactivates it. Subsequent research has shown that in the case of idronoxil, the second step is non-existent in many cancers. NOX66 has been designed to deliver idronoxil in a way that protects it from this 2-step process so that idronoxil reaches cancer cells in a fully active state.
A slightly longer answer:
In defence of those earlier studies, idronoxil (or phenoxodiol) was the first member of a family of chemicals known as isoflavones to be tested so extensively in humans. There simply wasn’t any textbook on their use; and there still isn’t.
That was pioneering work that came with a lot of unknowns. Not the least of those unknowns is what happens within the micro-environment of a cancer, where any anti-cancer drug faces a series of hurdles that start with leaving the bloodstream, finding its way to an individual cancer cell, penetrating the cell, and finding its molecular target in the cancer cell in spite of a range of attempts on the cancer cell’s part to avoid the drug.
We know considerably more about our universe than we do about the behaviour of a cancer cell. We are far more adept at landing a spacecraft on a distant planet and having it perform a range of sophisticated tasks than we are at delivering a drug to a cancer cell and guaranteeing that it will work.
Idronoxil runs two gauntlets in doing its job as an anti-cancer drug. The first is a detoxification process in the body known as Phase 2 metabolism which serves to inactivate the drug; the second is a reliance on the cancer cell to reactivate the drug, when that cell is doing everything in its power to defend itself and stop the reactivation.
I am not going to even try to explain Phase 2 metabolism in any great detail. It’s too complicated. Google it if you want to understand it. Basically it is a defence mechanism developed by the body to deal with foreign chemicals that enter it that are not soluble in water. It is important for the body to be able to eliminate foreign chemicals as quickly as possible, just in case they are poisonous. And the quickest method of elimination is through the kidneys. But that means being soluble in water (urine). Phase 2 metabolism is a process by which the body converts foreign chemicals (such as drugs) that are not soluble in water, into a form that is soluble in water, so that they can be processed through the kidneys.
The problem with that is that once the drug has been converted into this water-soluble form, it loses its ability to work. Regaining that ability means entering a cell, any cell, where specific enzymes are present that reactivate the drug. Once reactivated, the drug then is able to work.
Normal, healthy cells comply with this process by containing the necessary reactivation enzymes; cancer cells typically don’t contain enough of these reactivating enzymes, leaving idronoxil marooned in the cancer cell in an inactive state.
NOX66 has been designed to block idronoxil being subject to Phase 2 metabolism. No metabolism means that the idronoxil is not reliant on the cancer cell re-activating it, and thus is free to work.
A much longer answer
Probably the best way to understand what is going on is to follow the journey of a molecule of idronoxil once it enters the body.
Let’s start with the drug being taken orally.
Our idronoxil molecule is fairly readily absorbed by the lining of the stomach and small intestine and passed into the bloodstream. It is in the lining of the gut that the idronoxil molecule first encounters the front-line of the Phase 2 metabolism process. This is a family of enzymes (known as transferases) that attach various other molecules (glucuronic acid, sulphate) to the idronoxil molecule designed to make it water-soluble.
What passes from the lining of the gut into the bloodstream are 8 possible forms of idronoxil. The 8 possible forms come from the fact that the idronoxil molecule has two ends where glucuronic acid or sulphate can be attached. The following diagram shows the 8 possible combinations that end up in the bloodstream.
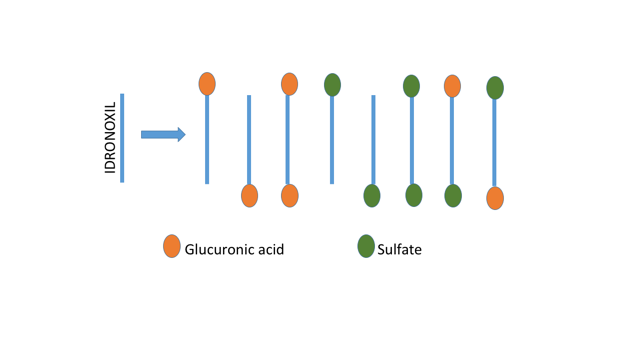
Any idronoxil that managed to get through the lining of the gut unscathed would be quickly picked up by the liver where there is an even higher level of Phase 2 enzymes ready to do the conversion into these 8 possible forms. The combined effects of the lining of the gut + liver ensuring that virtually all idronoxil in the body is now in these 8 forms.
Giving idronoxil intravenously instead of orally makes no difference to this outcome. Any drug injected intravenously has to pass through the liver which is very efficient at filtering out foreign chemicals such as idronoxil and subjecting them to Phase 2 metabolism. The end result is the same with either route of administration.
Our 8 different forms of idronoxil molecules now circulate freely in the bloodstream, coming in contact with every tissue in the body, not just cancer tissue. Idronoxil doesn’t discriminate – it will enter every cell it comes across. If our molecule enters a bone cell, for example, then the inside of that cell contains a collection of enzymes designed to reverse the Phase 2 metabolism. These are enzymes with a highly specific purpose – to remove glucuronic acid and sulphate from compounds with a chemical structure similar to idronoxil. In fact, there are 4 different enzymes required to do this task: one to remove glucuronic acid from the ‘A’ end of the idronoxil molecule and another from the ‘B’ end; similarly, two other enzymes to remove the sulphate group from the ‘A’ and ‘B’ ends.
Freeing idronoxil from its attached glucuronic acid or sulphate means that it now is in an active state and free to bind to its protein target. But healthy (non-cancer) cells don’t express this target, and so the idronoxil leaves the cell, re-enters the bloodstream, is filtered out by the liver and re-converted into one of the 8 water-soluble forms, and then passed out in the urine. A convoluted, but relatively uneventful journey.
The problem for idronoxil comes from being able to reproduce this process with a cancer cell. The target of idronoxil is a protein located on the outside of the cancer cell, which means that on reaching the cancer cell in its inactive state, it needs to enter the cancer cell, be converted back to an active state, exit the cancer cell, and locate and bind to its protein target sticking out of the cancer cell’s external surface.
What could possibly go wrong with this journey?
Well, to start with, cancer cells are not normal cells. They increase some functions and decrease others. It turns out that one of the functions they decrease is the 4 reactivation enzymes. That means that the idronoxil is marooned inside the cancer cell, doomed to remain stuck to its attached second molecule in an inactive state.
Then, just to add insult to injury, some cancer cells actually develop their own Phase 2 metabolism activity, so that even if the idronoxil could be freed, it would end up being re-attached just as quickly.
Both outcomes are part of a cancer cell’s determination to do whatever it takes to survive. In this case, working to ensure that an anti-cancer drug such as idronoxil is hobbled.
Why did idronoxil (phenoxodiol) work sometimes and not others?
Idronoxil underwent 10 years of clinical development, including going all the way into a Phase 3 registration study. That sort of commitment does not happen without some evidence of efficacy along the way. The problem appears to have been that such efficacy was inconsistent and unpredictable.
It appears to have nothing to do with earlier routes of administration. Both oral and intravenous routes of administration are reported to have produced clinical responses in late-stage cancer patients. A Phase 2 study in late-stage prostate cancer with oral idronoxil monotherapy delivered a dose-response effect; another Phase 2 study of intravenous idronoxil plus carboplatin delivered an unexpected 30% response rate in highly refractory late-stage ovarian cancers. So, this was not a case of oral versus intravenous.
I can only speculate. Take your pick from:
- Phase 2 metabolic processes are incomplete in some people, resulting in a proportion of idronoxil in the bloodstream remaining in an active form;
- Cancer cells vary in their ability to re-activate idronoxil;
- Cancer cells vary in their ability to conduct their own Phase 2 metabolism.
My guess is that all 3 are likely to be in play and accounting for the drug’s inconsistent and unpredictable effects to date.
The NOX66 technology
The technology was designed to take the whole issue of Phase 2 metabolism completely out of play and deliver idronoxil to the cancer cell in an active state where its ability to combine with its target was independent of any capacity of the cancer cell to defend itself.
The ‘light bulb’ moment for me came in early 2012 with the realisation that the highly potent anti-cancer effects that had been observed with idronoxil in mice was associated with low levels of Phase 2 metabolism in that species. In mice, only 20-30% of isoflavonoid compounds such as idronoxil are present in the blood in an inactive form; the remainder is there in an active form. That was in stark contrast with the virtually 100% level of Phase 2 metabolism in humans.
The pattern was pretty clear:
- In the test-tube where there was no Phase 2 metabolism, idronoxil displayed extraordinary potency, allowing cytotoxic drugs such as carboplatin to be lowered by thousands of times and still be capable of killing cancer cells;
- In mice where there was some (but incomplete) Phase 2 metabolism, idronoxil provided a significant anti-cancer effect in combination with drugs such as carboplatin;
- In humans where there is complete Phase 2 metabolism, the drug struggled to work.
The answer seemed to me to be to protect the drug from Phase metabolism. Some basic experimentation then established that wrapping the idronoxil in a chemical envelope effectively protected the drug from the body’s Phase 2 metabolic processes. That means that the idronoxil now reaches the cancer cell in an active form and is able to attach to its target on the outside of the cancer cell without having to enter the cell and face the uncertainty of what lies inside the cell.
The technology is the subject of a number of patent applications, so the precise details will remain confidential for the moment.