Our last announcement (27th March) reported on two developments. Both relating to some research being done under contract for Noxopharm by Monash University and partially funded by the Federal Government.
First development: idronoxil-C
Q. What is idronoxil-C?
A. Idronoxil-C is an exciting development that we believe has increased very considerably the value of the Company’s IP assets, opening up some potentially very important therapeutic and commercial opportunities.
When we dose with NOX66, the idronoxil is absorbed into the body and appears in the blood in various forms. This is something that happens with most drugs that humans take. In the case of idronoxil, some of these forms are inactive. But we have discovered one very distinctive form that we refer to as idronoxil-C that we believe is the main active form. This is a novel (and patentable) form of idronoxil.
Q. What is so exciting about discovering idronoxil-C?
A. Two main reasons. The first is that when it comes to seeking approval to market a new drug such a NOX66, understanding how the product is working helps the approval process considerably. It’s not essential to know everything about how a drug is working to get marketing approval, but it helps.
The second is that we have identified what essentially is a new drug. It’s still idronoxil, but it’s idronoxil in a form that makes it work better, and that opens up a wide range of opportunities.
From here we either can give patients idronoxil-C indirectly (via NOX66), or directly (pure idronoxil-C products). Our strategy is to develop both, with the indirect route (NOX66) our current focus.
Second development: urogenital cancers
Q. What are urogenital cancers?
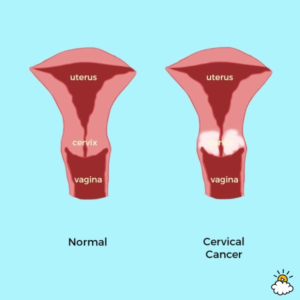
A. These are cancers of the female genital system (uterus, vagina, vulva, cervix) and of the bladder in both men and women.
Collectively these cancers are referred to as urogenital cancers, and each of them carries a poor outcome once it spreads from its site of origin.
Q. What are we aiming to do?
A. Our initial aim is to develop a specific formulation (to be called NOX77) to treat female genital cancers, focusing in the first instance on cancer of the cervix. This formulation will be based on idronoxil-C.
Q. What is cervical cancer?
A. Cervical cancer is cancer arising in the cervix (neck of the womb). If it is detected early enough (eg by PAP smear or physical examination) while it is still confined to the cervix, then that can be the end of the matter. The problem arises when it migrates away from the cervix. Starting with spreading to other tissues within the pelvis (bowel, bladder, lymph nodes, before then spreading beyond the pelvis. Chemotherapy and radiotherapy offer only limited benefit once the cancer had spread.
Q. How are we proposing to use NOX77?
A. Many women who go on to die from cervical cancer are diagnosed when the cancer still is contained within the pelvic cavity.
The purpose of NOX77 will be to help treat the cancer in this relatively early stage. The objective will be to concentrate idronoxil-C in the pelvic tissues where it will do the most good, with high levels of drug in the cancer tissue rendering the cancer cells far more responsive to chemotherapy and radiotherapy than they otherwise would be.
Q. Why not just use NOX66?
A. We have every reason to believe that NOX66 could do the same job. But NOX66 is designed for general use throughout the body, including the brain, with NOX66 delivering idronoxil-C fairly evenly throughout the body. And while idronoxil-C delivered by NOX66 is perfectly capable of seeking out the cervical cancer cells within the pelvic cavity, or anywhere else for that matter, it makes sense that if we can give idronoxil-C in a way that ensures that most of it passes through the pelvic tissues where it is needed the most, then that is a worthwhile strategy.
One product doesn’t nullify the other. In fact, using a combination of NOX66 and NOX77 would be an obvious approach, particularly where the cancer has spread beyond the pelvis, but where being able to concentrate the drug at the site of the cancer’s origin remains an important arm of the treatment.
Q. Isn’t cervical cancer becoming less common?
A. Yes, at least in many developed countries. The Australian-developed vaccine (‘Gardasil”) is making a difference. But the vaccine doesn’t work in all women, plus vaccination is limited to young women. Its cost also means it is limited to first-world countries.
About 250 women are expected to die from cervical cancer this year in Australia, and about 4200 women in the US. World-wide, cervical cancer remains a significant problem, with an estimated quarter of a million deaths expected this year. It remains a major problem worth solving.